Neuro Devices Market Research Report
Neuro Devices Market Research Report
Neuro Devices Market Opportunities & Risk Assessment
Global Neuro Devices Market is segmented by Application (Neurological Disorders, Brain Surgery, Pain Management, Epilepsy Treatment, Rehabilitation), Type (Neurostimulation Devices, Neuro Monitoring Devices, Neurosurgical Devices, Brain Computer Interfaces, Spinal Cord Stimulators), and Geography (North America, LATAM, West Europe, Central & Eastern Europe, Northern Europe, Southern Europe, East Asia, Southeast Asia, South Asia, Central Asia, Oceania, MEA)
Pricing
INDUSTRY OVERVIEW
The Neuro Devices is Growing at 9.30% and is expected to reach 92.60 billion by 2033. Below mentioned are some of the dynamics shaping the Neuro Devices.
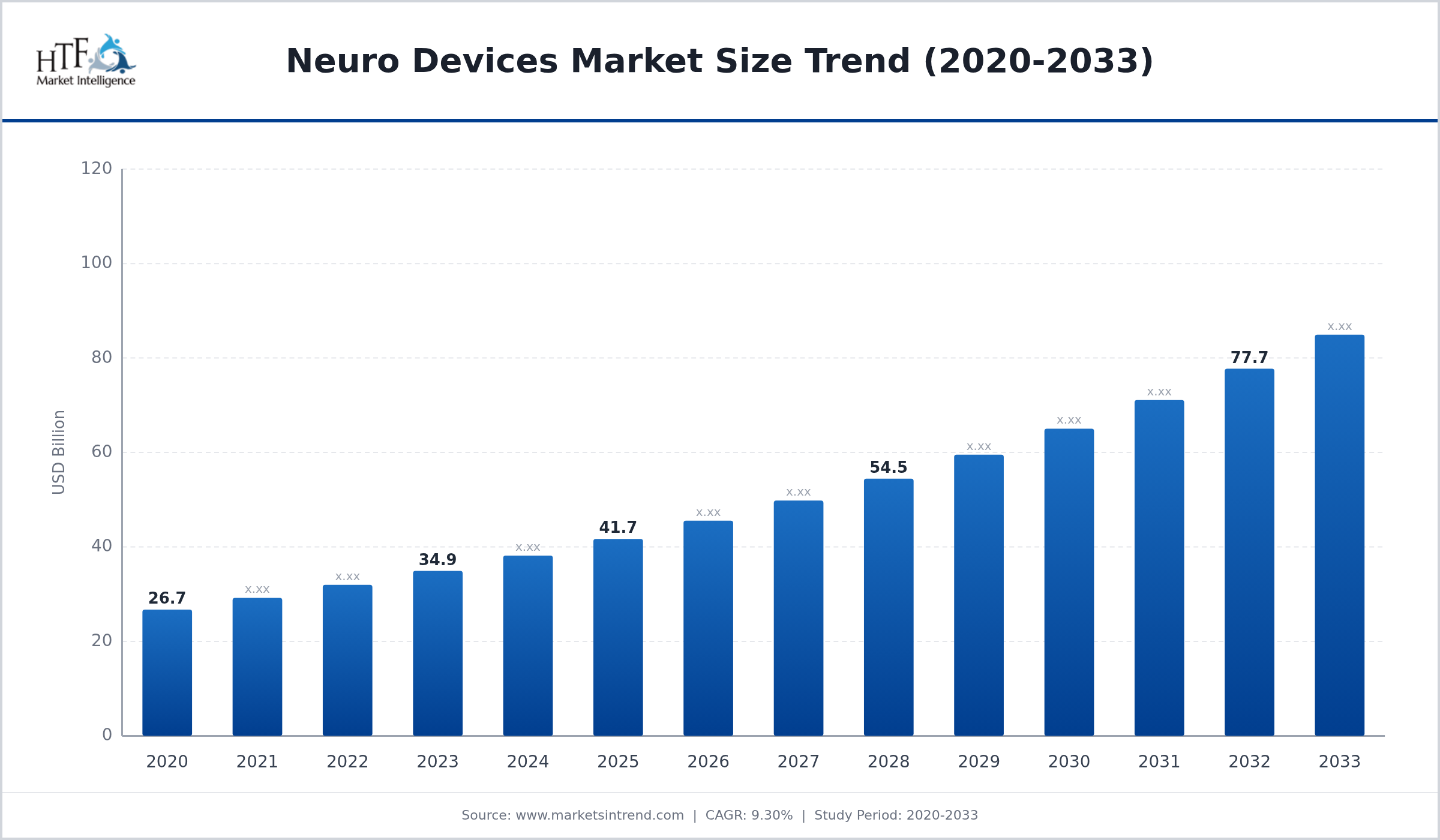
Neuro Devices Market refers to medical devices designed to diagnose monitor and treat neurological disorders affecting the brain spinal cord and nervous system. These devices include neurostimulators brain implants neuro-monitoring systems and diagnostic equipment used in neurology and neurosurgery. Neuro devices help manage conditions such as epilepsy Parkinson’s disease chronic pain and stroke. They improve patient outcomes by providing targeted treatment and monitoring capabilities. Advancements in neuroscience and increasing prevalence of neurological disorders are driving the neuro devices market globally.
Regulatory Landscape
- • Regulatory landscape emphasizes medical device approval clinical validation and safety standards requiring neuro devices to adhere to healthcare regulations ensuring patient safety globally
Market Drivers:
The key drivers in the market include technological advancements, increasing demand by consumers for innovative products, and government-friendly policies. Our research company combines industry reports with expert interviews and market analysis tools to identify and quantify drivers such as these. We review the current trends and gather data from leading industry publications and market research firms to decipher exactly how these and other factors are encouraging or dampening market growth.
- • Growth in neurodegenerative diseases
- • Chronic pain alternatives
- • Mental-health technology funding
- • Brain-computer interface R&D
- • Aging demographics
Some of the restraints to market growth may include regulatory challenges, high production costs, and disruptions in the supply chain. Our sources for these limitations include the regulation filings, industry surveys, and direct contributions from active participants within this marketplace. Tracking policy updates and economic reports further helps us to determine what kind of effect these factors have on the industry.
- • Surgical implantation risk
- • Cyber-security of implants
- • Long regulatory timelines
- • High device costs
- • Reimbursement variability
Among the trending ones are sustainability, digital transformation, and increasing importance of data analytics. Our research company is tracking these trends through the use of trend analysis tools, social media sentiment analysis, and industry benchmarking studies. Insights in emerging market preferences and technological advancements also come from surveys and focus groups.
- • Closed-loop deep-brain stimulators
- • Non-invasive vagus patches
- • Wireless EEG headbands
- • AI seizure-prediction software
- • Rechargeable implant batteries
These include emerging markets, innovation in product development, and strategic partnerships. We identify these opportunities by performing market segmentation analysis, competitive landscape assessment, and investment trend evaluation. The data is collected based on industry reports, financial performance analysis for major players, and forecasting models for identifying future growth areas.
- • Emerging-market clinics
- • Home therapy wearables
- • Data subscription for clinicians
- • Pharma combo therapies
- • Fast-track orphan indications
Regulation Shaping the Healthcare Industry
The healthcare industry is significantly influenced by a complex framework of regulations designed to ensure patient safety, efficacy of treatments, and the overall quality of care. Key regulatory areas include drug approval processes, medical device standards, and healthcare data protection. These regulations aim to maintain high standards for clinical practices and safeguard public health.
Major Regulatory Bodies Worldwide
1. U.S. Food and Drug Administration (FDA): In the United States, the FDA is a pivotal regulatory authority overseeing the approval and monitoring of pharmaceuticals, medical devices, and biologics. The FDA sets stringent standards for product safety and efficacy, which significantly impacts market entry and ongoing compliance for healthcare companies.
2. European Medicines Agency (EMA): The EMA plays a crucial role in the European Union, evaluating and supervising medicinal products. It provides centralized approval for drugs and ensures that products meet rigorous safety and efficacy standards across member states.
3. Health Canada: This agency regulates pharmaceuticals and medical devices in Canada, ensuring that products are safe, effective, and of high quality. Health Canada's regulations are aligned with international standards but tailored to meet national health needs.
4. World Health Organization (WHO): While not a regulatory body in the traditional sense, the WHO sets international health standards and provides guidelines that influence national regulatory frameworks. It plays a key role in global health policy and emergency response.
5. National Medical Products Administration (NMPA): In China, the NMPA regulates the approval and supervision of drugs and medical devices, with an increasing focus on aligning with global standards and facilitating market access.
SWOT Analysis in the Healthcare Industry
SWOT analysis in the healthcare industry involves a structured assessment of Strengths, Weaknesses, Opportunities, and Threats to identify strategic advantages and areas for improvement.
• Strengths: Evaluates internal factors such as advanced technology, skilled personnel, and strong brand reputation. For example, a hospital with cutting-edge medical equipment and specialized staff is considered to have a strong competitive edge.
• Weaknesses: Identifies internal limitations like outdated facilities, regulatory compliance issues, or high operational costs. Weaknesses could include inefficient processes or lack of innovation.
• Opportunities: Assesses external factors that could drive growth, such as emerging medical technologies, expanding markets, or favorable government policies. Opportunities might involve partnerships or new service lines.
• Threats: Examines external challenges such as increasing competition, changing regulations, or economic downturns. Threats might include new entrants with disruptive technologies or stricter regulatory requirements.
Market Segmentation
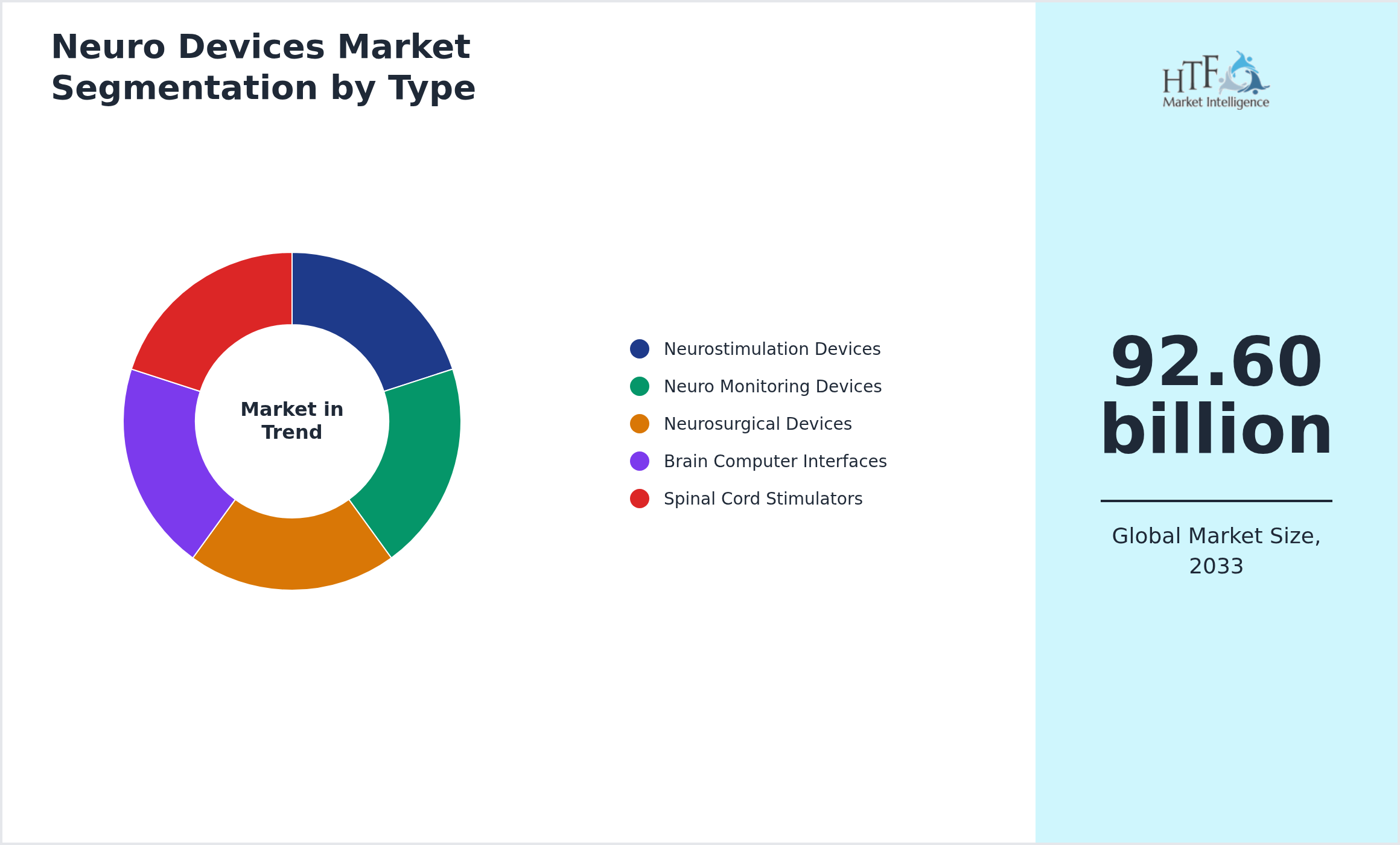
Segmentation by Type
- • Neurostimulation Devices
- • Neuro Monitoring Devices
- • Neurosurgical Devices
- • Brain Computer Interfaces
- • Spinal Cord Stimulators
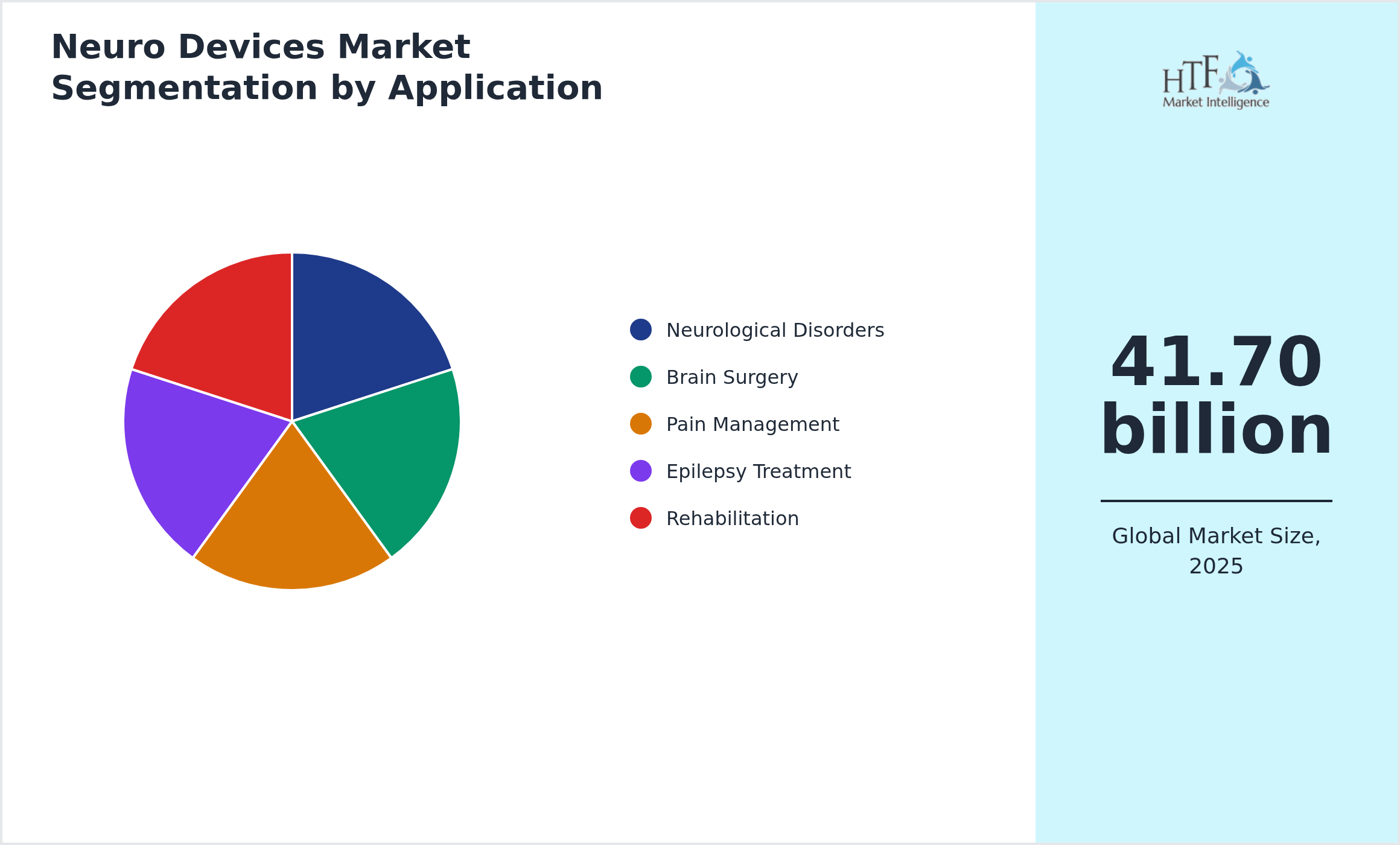
Segmentation by Application
- • Neurological Disorders
- • Brain Surgery
- • Pain Management
- • Epilepsy Treatment
- • Rehabilitation
Regional Outlook
The North America currently holds a significant share of the market, primarily due to several key factors: increasing consumption rates, a burgeoning population, and robust economic momentum. These elements collectively drive demand, positioning this region as a leader in the market. On the other hand, Asia Pacific is rapidly emerging as the fastest-growing area within the industry. This remarkable growth can be attributed to swift infrastructure development, the expansion of various industrial sectors, and a marked increase in consumer demand. These dynamics make this region a crucial player in shaping future market growth. In our report, we cover a comprehensive analysis of the regions and countries, including
- North America
- LATAM
- West Europe
- Central & Eastern Europe
- Northern Europe
- Southern Europe
- East Asia
- Southeast Asia
- South Asia
- Central Asia
- Oceania
- MEA
The company consistently allocates significant resources to expand its research capabilities, develop new medical technologies, and enhance its pharmaceutical portfolio. Johnson & Johnson's investments in R&D, coupled with strategic acquisitions and partnerships, reinforce its position as a major contributor to advancements in healthcare. This focus on innovation and market expansion underscores the critical importance of the North American region in the global healthcare landscape.
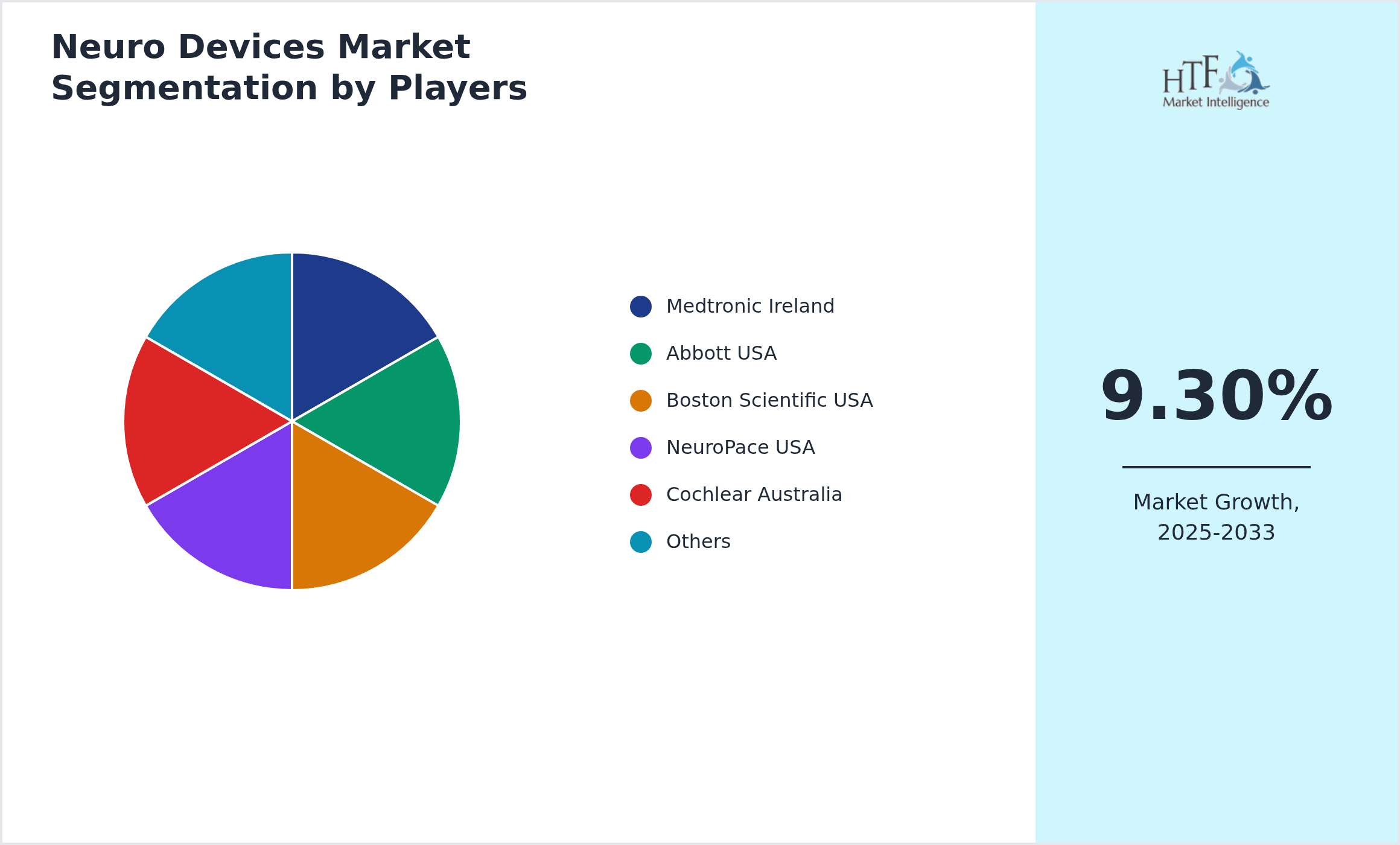
- • Medtronic Ireland
- • Abbott USA
- • Boston Scientific USA
- • NeuroPace USA
- • Cochlear Australia
- • LivaNova UK
- • Siemens Healthineers Germany
- • GE Healthcare USA
- • Philips Netherlands
- • Stryker USA
- • Johnson And Johnson USA
- • Natus Medical USA
- • Elekta Sweden
- • Brainlab Germany
- • Nihon Kohden Japan
Regional Analysis
- • North America dominates due to advanced healthcare infrastructure and strong R&D investments Europe follows with regulatory approvals and innovation Asia Pacific fastest growing driven by rising neurological disorders and expanding healthcare access across emerging markets
Market Entropy
- • Jan 2026: Advanced neurostimulation and brain-monitoring devices released for neurological disorder management and rehabilitation.
Merger & Acquisition
- • Feb 2026 – NeuroLink Medical acquired BrainPulse Technologies to expand implantable stimulation systems. Sep 2025 – GlobalNeuroTech Group merged with Synaptech Solutions to strengthen epilepsy and Parkinson’s device pipeline.
Regulatory Landscape
- • Regulatory landscape emphasizes medical device approval clinical validation and safety standards requiring neuro devices to adhere to healthcare regulations ensuring patient safety globally
Patent Analysis
- • Medtronic develops patents in neuro devices improving treatment outcomes Abbott focuses on implantable technologies enhancing functionality Boston Scientific innovates in neuromodulation strengthening capabilities Nevro expands intellectual property in neuro stimulation improving performance LivaNova advances patents enabling advanced neurological treatment technologies globally
Investment and Funding Scenario
- • Increasing investments in neuroscience and medical technology drive funding in neuro devices markets enhancing innovation globally Strategic partnerships between medical device companies and research institutions accelerate adoption and development Government initiatives supporting healthcare promote deployment of advanced neurological solutions across regions Private equity investments enable expansion of manufacturing capabilities improving scalability globally Rising demand for neurological treatments attracts funding supporting market growth globally
Primary and Secondary Research
Primary research involves the collection of original data directly from sources in the healthcare industry. Approaches include the survey of health professionals, interviews with patients, focus groups, and clinical trials. This gives an overview of the current practice, the needs of the patient, and the interest in emerging trends. Firsthand information on the efficacy of new treatments, an assessment of market demand, and insight into changes in regulation can be sought only with primary research.
Secondary Research: This is the investigation of existing information from a variety of sources, which may include industry reports, academic journals, government publications, and market research studies. Alfred secondary research empowers them to understand trends within industries, historical data, and competitive landscapes. It gives a wide view of the market dynamics and validates findings obtained from primary research. By combining both primary and secondary together, health organizations will be empowered to develop comprehensive strategies and make informed decisions based on a strong foundation built on data.
Report Infographics
|
Report Features |
Details |
|
Base Year |
2025 |
|
Based Year Market Size (2023) |
41.70 billion |
|
Historical Period |
2020 to 2025 |
|
CAGR (2025 to 2033) |
9.30% |
|
Forecast Period |
2025 to 2033 |
|
Forecasted Period Market Size (2033) |
92.60 billion |
|
Scope of the Report |
|
|
Regions Covered |
North America, LATAM, West Europe, Central & Eastern Europe, Northern Europe, Southern Europe, East Asia, Southeast Asia, South Asia, Central Asia, Oceania, MEA |
|
Companies Covered |
Medtronic Ireland, Abbott USA, Boston Scientific USA, NeuroPace USA, Cochlear Australia, LivaNova UK, Siemens Healthineers Germany, GE Healthcare USA, Philips Netherlands, Stryker USA, Johnson And Johnson USA, Natus Medical USA, Elekta Sweden, Brainlab Germany, Nihon Kohden Japan |
|
Customization Scope |
15% Free Customization (For EG) |
|
Delivery Format |
PDF and Excel through Email |
